Introduction
Recognising problems in the experimental setup is important. Drift, jumps and spikes in the curves make analysing sensorgrams difficult. Analysing suboptimal sensorgrams will lead to erroneous results and waste experimental time. Therefore it is vital to identify problems in the experimental set-up and solve them before analysing is attempted. This part will high light several problems and give some hints to diagnose where the problem is and provide solutions to optimize the experiment.
Fresh buffers
Ideally fresh buffers are prepared each day and 0.22 µM filtered and degassed before use. We normally make 2 litres of buffer and 0.22 µM filter. Storage is in clean (sterile) bottles at room temperature. Keep in mind that buffers stored at 4°C contain more dissolved air which can create air-spikes in the sensorgram. Just before use, transfer an aliquot of the buffer to a new clean bottle and degass. It is bad practice to add fresh buffer to the old since all kind of nasty things can happen / growing in the old buffer. After degassing, add a detergent when suitable. A good running buffer hygiene is step one for better results.
Baseline drift
Baseline drift is usually a sign of non-optimal equilibrated sensor surfaces. Drift is often seen directly after docking a (new) sensor chip or after the immobilization of the sensor surface. This is due to the rehydration of the surface and the wash-out of chemicals used during the immobilization procedure or the adjustment of the bound ligand to the flow buffer. It can be necessary to run the running buffer overnight to equilibrate the surfaces.
Drift can also occur after a change in running buffer. In general, prime the system after each buffer change and wait for a stable baseline. Failing to equilibrate the system will result in a waviness pump stroke because the previous buffer is mixing with the next buffer in the pump. After some pump strokes the signal will be stable again.
Start-up drift can be seen when the flow is initiated after a flow stand still. Some sensor surfaces are susceptible to flow changes and this will be visible as a drift that will level out over time (5–30 minutes). The duration of this effect depends on the type of sensor and the ligand bound to it. It is advised to wait for a stable baseline before injection of the first sample. In systems where this is not possible, a short buffer injection and a five minute dissociation time can stabilize the baseline before the analyte injection is done. Regeneration solutions can also have an effect on the drift. This can be different between the reference and active surface due to the difference in protein and immobilization level. When using long dissociation times, equal drift rates between channels must be established or double referencing must compensate for drift differences sufficiently. To equilibrate the system, use sufficient buffer in prime and wash steps. Flow the running buffer at the flow rate of the experiment until a stable baseline is obtained.
Equilibration, baseline and noise level
Equilibration of the system is done by flowing running buffer over the sensor surfaces and monitoring the baselines. Start with preparing sufficient buffer for the experiment and filter and degas the solution. Add the appropriate detergent after filtering and degassing to avoid foam forming. Change the buffer in the pumps and tubing by priming the system several times or by flowing the buffer through the system.
To determine the noise level of the instrument, first equilibrate the system to minimize drift. Then inject running buffer several times and observe the average baseline response.
In the figure ‘Buffer injection’, a 5 minute equilibration time is set before the injection is started (1). When the injection needle hits the injection port (2), the baseline drops an average of 2 RU's. The pump fill (3) stops the flow for some seconds and a spike in the system occurs. At point (4), the buffer injection starts and it ends at point (5).
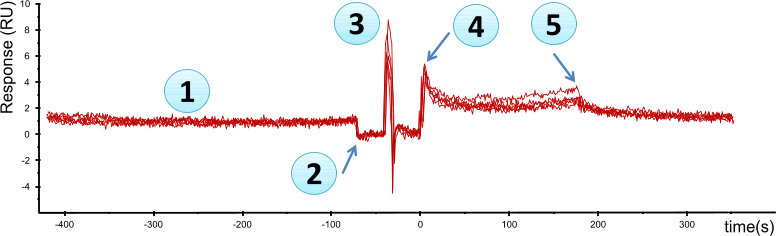
The system is very sensitive to pressure differences, which cause abrupt response changes. Nevertheless, the overall noise level is very low (< 1 RU). If one of the curves is used as a blank, the overall signal will be < 1 RU even during the buffer injection.
The average response level is the starting point for the next experiments. In addition, check the shape of the curves. If there is drift or the curves are not level shortly after the injection start, equilibrate better or clean the instrument. If the response between the flow channels is not comparable, it may be an indication that the IFC or sensor needs replacement (1) or that the detector should be recalibrated.
Therefore, it is advised to use a steady running buffer flow and incorporate several dummy injections (running buffer) with regeneration at the start of an experiment to stabilize the system. In addition, in systems where this is not possible, a short buffer injection and a five minute dissociation time can stabilize the baseline.
Experimental set-up
A proper experimental set-up can minimize baseline drift. This starts with a prime of the system after each buffer change and start of a method. Extra time to equilbrate the system is required after cleaning the system.
Add start-up cycles
In the experimental method, add at least three start-up cycles. These cycles are the same as the cycles with analyte but inject buffer instead of analyte. If a regeneration step is required, the regeneration injection is also done. In this way the surface is 'primed' and possible differences induced by the first regeneration cycles are left out of the experiment. The start-up cycles are left out of the analysis of the experiment. Do not use them as blanks.
Add blank injections
Add some blank (buffer alone) cycles in the method. It is better too have to much than too few blank cycles. It is recommanded to add an average of one blank cycle every five to six analyte cycles and end with one. Blank cycles makes it possible to perform double referencing (see below).
Double referencing
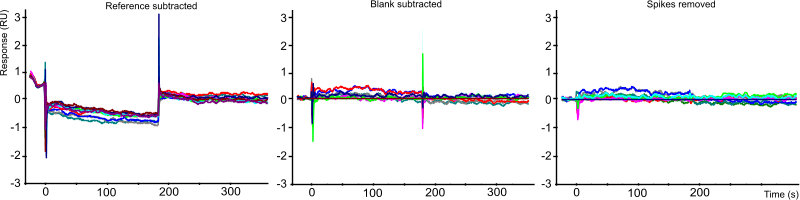
Double referencing is the procedure to compensate for drift, bulk effect and channel differences. First a reference (negative) channel is subtracted from the active channel. This will compensate for the main bulk effect and drift. Then the blanks (running buffer only) are subtracted. This will compensate for differences between the reference and active channel. To have the best referencing the reference channel should closely match the active channel. Use several blanks during the measurement and space the blanks evenly within the experiment.
References
| (1) | Myszka, D. G. Improving biosensor analysis. J.Mol.Recognit. 12: 279-284; (1999). Goto reference |
Footnotes
- Also read the Bulk and Spikes topic.